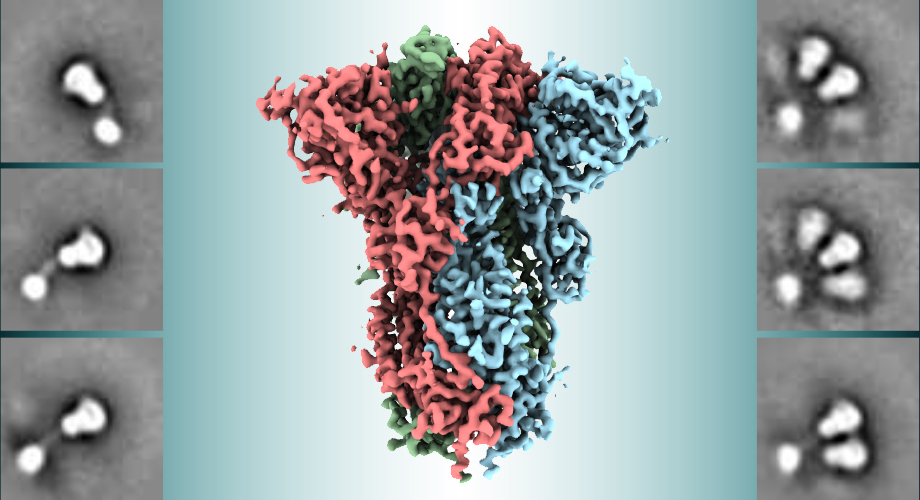
Electron microscopy techniques showed that the coronavirus spikes maintained the desired shape in the vaccine, which should help stimulate an appropriate immune response.
LA JOLLA, CA—Scientists at Scripps Research have obtained high-resolution, atomic-scale details of the structure of a SARS-CoV-2 “spike” protein from an experimental COVID-19 vaccine—details that are consistent with the robust neutralizing antibody responses the vaccine elicited in preclinical and phase 1 clinical studies.
The experimental vaccine, NVX-CoV2373, is being developed and tested in clinical trials by the vaccine maker Novavax Inc. It uses lab-grown copies of the SARS-CoV-2 spike protein to stimulate neutralizing antibody responses to this key viral protein, with the goal of blocking new COVID-19 infections.
The new structural details, obtained using low-temperature electron microscope techniques, are available in the manuscript submitted to the open-access preprint repository bioRxiv. The molecular map of the Novavax spike protein could be helpful in evaluating the vaccine’s ongoing human clinical trial results.
“Our findings suggest that the speedy design and manufacture of this vaccine did not compromise the quality of the spike protein structure,” says the study’s senior author Andrew Ward, PhD, a professor in the Department of Integrative Structural and Computational Biology at Scripps Research.
SARS-CoV-2, the virus that causes COVID-19, appears to have originated from a bat coronavirus in China. It has now caused nearly 20 million human infections globally, including more than 700,000 deaths. A vaccine to prevent infection is the world’s highest public health priority—and in pursuit of that goal, dozens of research teams have designed and started testing potential vaccines.
The Novavax vaccine, like most other COVID-19 vaccine candidates, is designed to stimulate an immune response—ideally providing long-term immune “memory” against the SARS-CoV-2 spike protein. These spike proteins stud the surface of each coronavirus particle; they are the protrusions that give the virus its crown-like appearance and contain the segments the virus uses to penetrate and then infiltrate host cells.
In what may be a strategy for evading immune recognition, the spike protein has a floppy, ever-changing shape. The Novavax immunogen spike protein uses mutations to stabilize the spike protein in its functional shape so that the immune system can learn to recognize and attack it. This key advancement is based on discoveries made several years ago by Ward and collaborating researchers Jason McLellan, PhD (University of Texas at Austin), and Barney Graham, MD, PhD (Vaccine Research Center at the National Institutes of Health) in their structural studies of the seasonal coronavirus HKU1 and of the pandemic coronaviruses MERS and SARS.
The new structural study shows that the mutations stabilize the Novavax spike protein in the desired conformation.
“It is incredibly gratifying to see how our prior work having direct impact on vaccine development for SARS-CoV-2,” Ward says. “The opportunity to characterize the vaccine formulation that Novavax is testing in people brings it full circle.”
“We are grateful for Andrew Ward and the scientists at Scripps Research for defining the high-resolution structure of the SARS-CoV-2 spike protein in our NVX-CoV2373 vaccine,” says Gale Smith, PhD, vice president of vaccine development and chief scientist at Novavax. “This important contribution provides insights into the high rate of transmission of COVID-19 and a scientific basis for the vaccine inducing neutralizing antibodies in 100 percent of phase 1 trial participants.”
Novavax has submitted data from the phase 1 clinical trial to a peer-reviewed scientific journal and the online preprint server medRxiv.org.
Ward notes that the structural details also indicate that the coronavirus spike protein has previously undiscovered “pockets” where certain compounds could bind. “These are places where, in principle, you could target the spike protein with small-molecule drugs,” he says.
The coronavirus spike proteins, like those of other viruses including HIV, naturally form three-copy structures called trimers on virus particles, and function in that trimer shape. Ward and his team, in the new study, found that the spike proteins not only form trimers successfully, but can also form clusters of two or three trimers.
The specific molecular nature of interactions points to the possibility—which Ward and colleagues can now follow up in further studies—that clustering of spike trimers occurs naturally on SARS-CoV-2 to enhance its infectivity.
“It’s possible that the virus needs more than one spike trimer to pierce a host cell, so this tendency to form higher-order clusters may underlie its infectiousness,” Ward says.
The study, “Structural analysis of full-length SARS-CoV-2 spike protein from an advanced vaccine candidate,” was authored by Sandhya Bangaru, Gabriel Ozorowski, Hannah Turner, Aleksandar Antanasijevic, Deli Huang, Xiaoning Wang, Jonathan Torres, Jolene Diedrich, John Yates III, David Nemazee, James Paulson and Andrew Ward, all of Scripps Research; and Jing-Hui Tian, Alyse Portnoff, Nita Patel, Michael Massare, Greg Glenn and Gale Smith of Novavax Inc.
Support was provided by the National Institute of Allergy and Infectious Diseases (UM1 AI144462, R01 AI113867, R01 AI132317), the Bill and Melinda Gates Foundation (OPP1170236) and Novavax Inc.